Filters
Question type
A) True
B) False
B) False
Correct Answer

verified
Correct Answer
verified
Question 22
Multiple Choice
Which of the following statements relating to the diagram of CO are true? Please select all that apply.
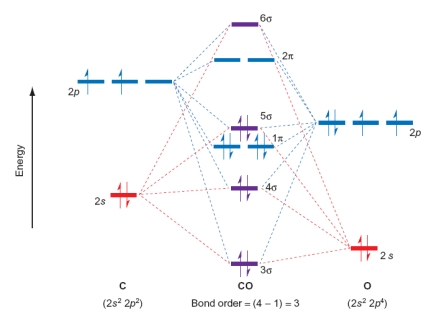
A) Adding electrons to form an anion will lower the bond order.
B) CO is paramagnetic.
C) Adding electrons to form an anion will increase the bond order.
D) CO is diamagnetic.
E) C) and D)
F) All of the above
F) All of the above
Correct Answer

verified
Correct Answer
verified
Question 23
Short Answer
A σ (sigma) orbital has __________ symmetry about the internuclear axis.
Correct Answer

verified
Correct Answer
verified
Question 24
Multiple Choice
How do the bond enthalpies and bond lengths for the homonuclear diatomics, H2, Li2, Na2 and K2 change as the group is descended?
A) Bond lengths increase and bond enthalpies increase.
B) Bond lengths increase and bond enthalpies decrease.
C) Bond lengths decrease and bond enthalpies increase.
D) Bond lengths decrease and bond enthalpies decrease.
E) B) and C)
F) A) and D)
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Question 25
Short Answer
According to Lewis theory, a nitrogen atom in N2 must share _ electrons to form a stable configuration.
Correct Answer

verified
Correct Answer
verified
Question 26
Short Answer
For a homonuclear diatomic, the bond length is _____the distance of the atomic radius.
Correct Answer

verified
Correct Answer
verified
Question 27
Multiple Choice
Taking z as the internuclear axis, how would the orbital formed from the bonding combination of two dz2 orbitals be labelled?
A) σu
B) σg
C) πg
D) πu
E) B) and C)
F) A) and D)
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 21 - 27 of 27
Related Exams